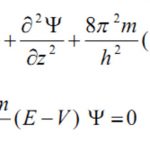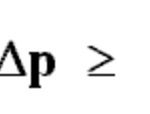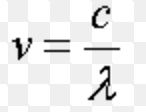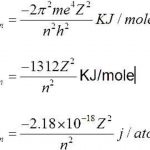Quantum mechanics, as developed by Erwin Schrodinger in 1926, is based on the wave motion associated with the particles. For the wave motion of the electron in the three dimensional space around the nucleus, he put forward an equation known as Schrondinger wave equation. where ψ is the amplitude of the wave where the coordinates of the electrons are ( x,y,z) ,E is the … [Read more...] about Quantum Mechanical Model of an Atom
Structure of Atom
Heisenberg’s Uncertainty Principle
Werner Heisenberg, a German physicist, in 1927 gave a principle about the uncertainty in simultaneous measurement of position and momentum of small particles. Heisenberg's uncertainty Principle states that: It is impossible to measure simultaneously the position and momentum of a small particle with absolute accuracy or certainty. The product of the uncertainty in the … [Read more...] about Heisenberg’s Uncertainty Principle
Dual Nature of Matter and Radiation
Einstein in 1905 suggested that light has dual nature, i.e. wave nature as well as particle nature. Matter wave or a de Broglie wave Louis de Broglie, a French physicist, in 1924, suggested that all microscopic as well as macroscopic objects possesses dual character. The wave associated with the particle is called a matter wave or a de Broglie wave. The wavelength of the … [Read more...] about Dual Nature of Matter and Radiation
Hydrogen Spectrum
Hydrogen Spectrum Atomic spectrum of hydrogen consists of a number of lines which have been grouped into 5 series :Lyman, Balmer, Paschen, Brackett and Pfund. Any given sample of hydrogen gas gas contains a large number of molecules. When such a sample is heated to a high temperature or an electric discharge is passed, the hydrogen molecules splits into hydrogen … [Read more...] about Hydrogen Spectrum
Bohr’s Model of an Atom
Neils Bohr, a Danish physicist in 1913 proposed a new model of atom. This new model is called Bohr's Model of Atom. Postulates of Bohr's model of an Atom 1) An atom consists of a small, heavy positively charged nucleus in the centre and electrons revolve around it in circular orbit. 2) The electrons revolve only in those orbits which have a fixed value of energy. These … [Read more...] about Bohr’s Model of an Atom