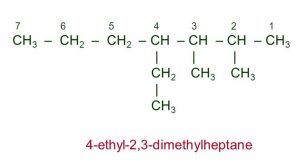
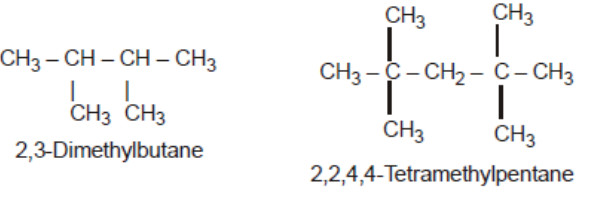
Longest chain rule: Select the longest continuous chain of carbon atoms. This is called the parent chain while all other carbon atoms which are not included in the parent chain are called branch chain or side chains or substituent.it may be noted that the longest chain may or may not be straight but it must be continuous.
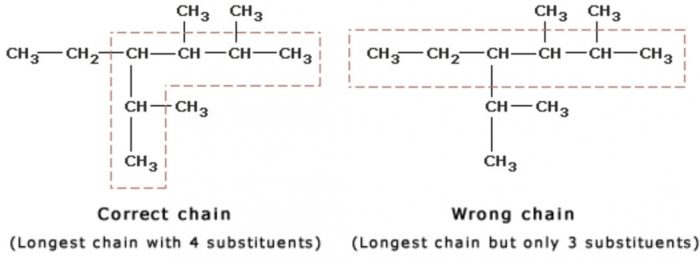
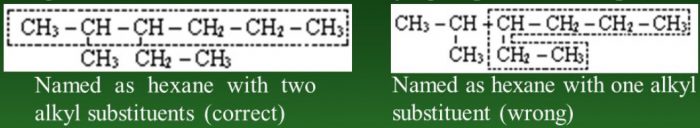
2) Rule for larger number of side chain : If two chains of equal length are possible, select the one with the larger number of side chains.
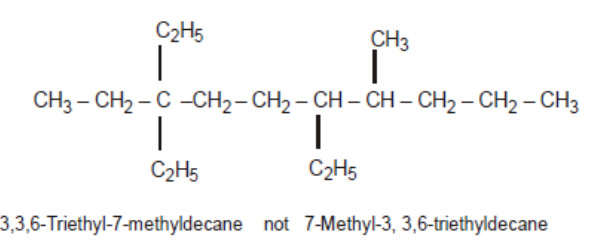
3) Lowest number rule : Number the carbon atoms as the parent chain as 1,2,3,4 …… etc starting from that end which gives the lowest possible number to the carbon atoms carrying the substituent.
The number that indicates the position of the substituent on the parent chain is called the positional number or the locant.
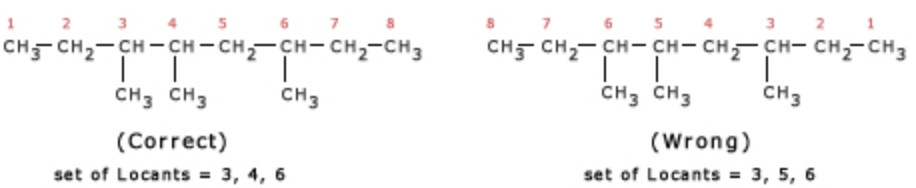
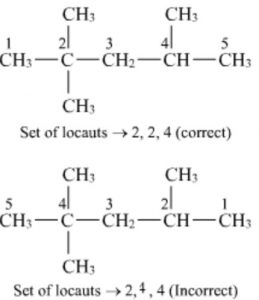
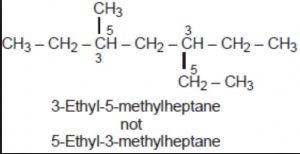
Lowest set of locant rule: When two or more substituents are present , the lowest set of locant rule is applied.When two or more different set of locants containing the same number of terms is possible , then that set of locant is the lowest which when compared term by term with other sets , each in order of increasing magnitude, has the lowest term at the first point of difference.
For deciding the lowest set of locants, the carbon atoms of the parent chain are numbered from all possible directions and a locant assigned to each substituent from each direction. The set of locants from each directions is then compared term by term till the first point of difference is reached. The set of locants is preferred which has a lower number at the first point of difference.
4) Name of the branched chain alkane: Prefix the name of the substituent to the name of the parent alkane and indicate its position by writing before it the number of carbon atom carrying the substituent.The name of the substituent is separated from its locant by a hyphen.The final name of the alkane is always written as one word.
5) Alphabetical order of the side chains: When two or more alkyl groups are present on the parent chain ,each alkyl group prefixed by its positional number is arranged in alphabetical order before the name of the parent alkane.
While deciding the alphabetical order of the various alkyl groups, prefixes iso and neo are considered to be part of the fundamental name of the alkyl group while the prefixes sec and tert are not.
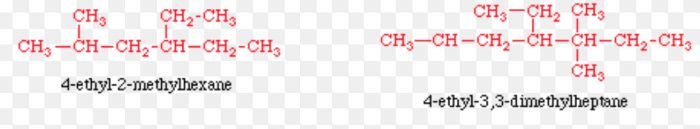
6) Numbering of different alkyl groups at equivalent positions: If two different alkyl groups are present at equivalent positions, the numbering of the parent chain is done in such a way that alkyl group which comes first in the alphabetical order gets the lowest number.
7) Naming same alkyl groups at different positions : When the same alkyl group occurs more than once on the parent chain at different positions, the positional number of each alkyl group is separated by commas and suitable prefixes such as di , tri, tetra are attached to the name of the alkyl group. However , the prefixes di, tri are not considered while deciding the alphabetical order of the alkyl group.
8) If same alkyl group occurs twice on the same carbon atoms, its positional number is also repeated twice.
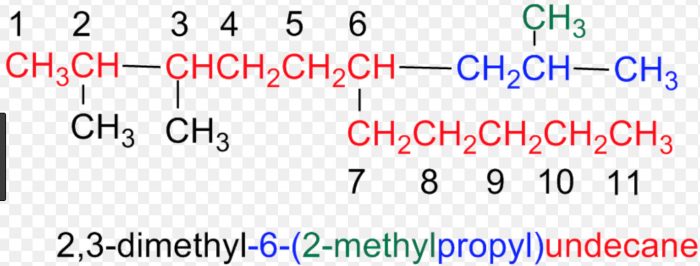
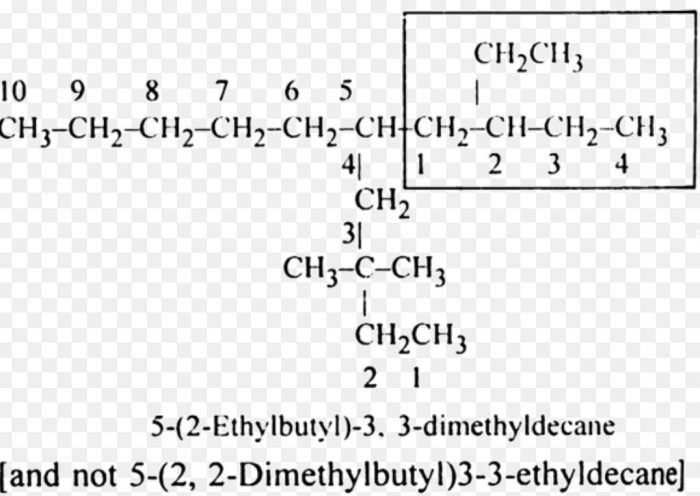
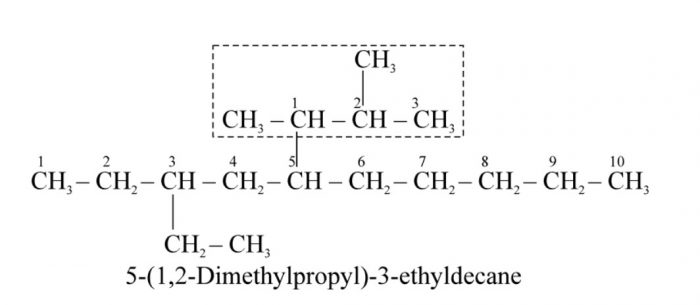
9) Numbering the complex substituent : In case the substituent on the parent chain is complex , it is named as a substituent alkyl group by numbering the carbon atoms of this group attached to the parent chain as 1.
The name of such a substituent is always enclosed in brackets to avoid confusion with the numbers of the parent chain.
10) If two complex substituents are of equal length , then the complex substituent with larger number of alkyl groups forms a part of the longest carbon chain while the other one is considered the real complex substituent.
11) While deciding the alphabetical order of the various substituent , the name of the complex substituent is considered to begin with the first letter of the complex name.
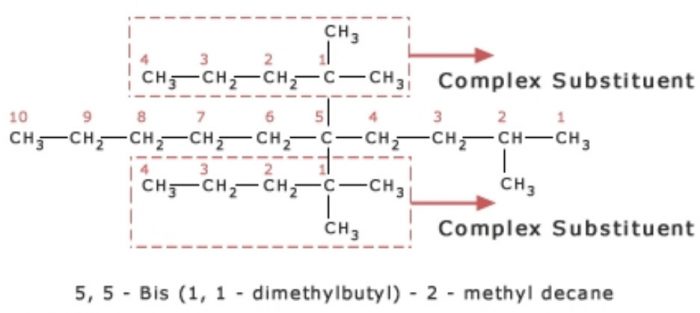
12) If the same complex substituent occurs more than once on the parent chain at different positions, prefixes bis , tris , tetrakis, pentakis are used before the name of the complex substituent.
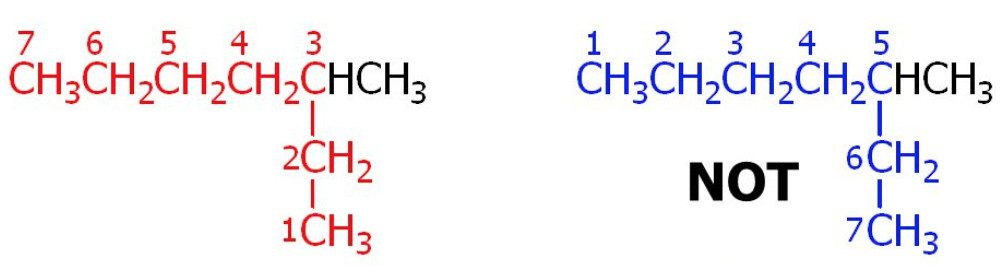
thanks lot
well and good but how can we down load it or send to other person
Thank you ma’am
thank you. it helped a lot ma’am
Thanks you, very nice example for preparing of questions longest chain And IUPAC Naming