Contents
Vapour Pressure of Liquid
When a liquid is allowed to evaporate in a closed vessel, part of the liquid evaporates and fills the available space with the vapours. Since the vapours leave the container, these get collected in the vapour state above the surface of the liquid.
Due to vaporisation, liquid changes into vapours and level of liquid decreases. As the evaporation proceeds, the number of gaseous molecules in the vapour phase increases gradually.
These molecules move about at random in a limited space during their random movement, some of these strike the surface of liquid and get condensed. The process of condensation acts in opposite direction to the process of evaporation. Thus, both evaporation and condensation processes go on simultaneously.
A stage is reached when the rate of evaporation becomes equal to rate of condensation and an equilibrium gets established between liquid and vapour phases.
The pressure exerted by the vapours above the liquid surface in equilibrium with the liquid at a given temperature is called vapour pressure.
Factors on which vapour pressure of a liquid depends
1) Nature of the liquid
Each liquid has a characteristic vapour pressure because each liquid has different magnitude of intermolecular forces.
The liquids, which have weaker intermolecular forces, tend to escape readily into vapour phase and therefore, have greater vapour pressure.
For example: dimethyl ether and alcohol have higher vapour pressure than water at a given temperature because of weaker intermolecular forces in them as compared to water.
2) Temperature
The vapour pressure of a liquid increases with increase in temperature. This is due to the fact that with increase in temperature, more molecules will have larger kinetic energies. Therefore, larger number of molecules will escape from the surface of the liquid to the vapour phase resulting in higher vapour pressure.
(a) Vapour Pressure of Liquid-Liquid Solution
When a binary solution of two volatile liquids is placed in a closed vessel, both the components would evaporate and eventually an equilibrium would be established between vapour phase and the liquid phase.
The French chemist Francois Marte Raoult (1886) gave a quantitative relationship between the partial pressure and the mole fractions of two components. This relationship is known as Raoult’s law which states that
at a given temperature, for a solution of volatile liquids, the partial vapour pressure of each component in solution is equal to the product of the vapour pressure of the pure component and its mole fraction.
Consider a binary solution of two volatile liquids and denote the components as A and B having the mole fraction xA and xB . If pA and pB are the vapour pressures of the components in the solution, then according to Raoult’s law
pA =pA° xA
pB =pB° xB
where pA° is the vapour pressure of the pure component A and pA° is the vapour pressure of pure component B at the same temperature.
According to Dalton’s law of partial pressures, the total pressure p over the solution phase in the container will be the sum of the partial pressure of the components of the solution.
This is given as
p=pA + pB
Substituting the values of pA and pB we get
p= pA° xA + pB° xB
xA + xB = 1
xA =1-xB
p= pA°(1- xB) + pB° xB
p= pA° – pA° xB + pB° xB
p= pA° – xB ( pB° – pA° )
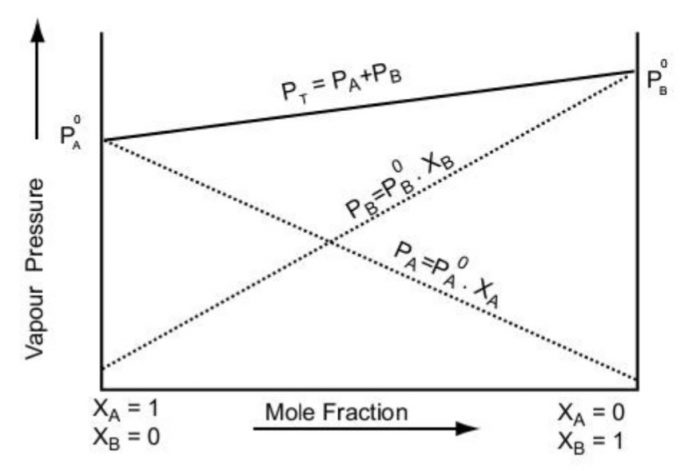
a) Total vapour pressure over the solution can be related to the mole fraction of any one component (xB or xA(1-xB))
b) Total vapour pressure of the solution varies linearly with the mole fraction of component B because pA° and pB° are constant.
c) Depending on the vapour pressure of the pure components A and B, total vapour pressure over the solution decreases or increases with the increase of the mole fraction of the component A.
The total vapour pressure (p) is a linear function of the mole fraction xB (or xA as xA =1-xB ) because pA° and pB° are constant at a particular temperature.
The dotted lines give the partial pressure of the two components versus composition and the solid line gives the vapour pressure versus composition.
According to Raoult’s law, the partial vapour pressures of two components A and B of a solution are given as:
pA = pA° xA
pB = pB° xB
Therefore, the vapour pressures of the components are linear functions of their mole fractions. Now
1) when xA=1, i.e., the liquid is pure A
pA =pA° × 1 = pA°
2) when xA = 0, i.e., the liquid is pure B
pA =pA° × 1 = 0
Thus, the vapour pressure of such solutions is either higher or lower than that predicted by Raoult’s law.
Thus, the plot of p against xA should give a straight line passing through pA° (when xA=1) and 0 (when xA = 0)
When the liquid is pure (xA =1), its vapour pressure is equal to pA°. As component B is added to component A (xA decreases), the vapour pressure decreases till it becomes zero (xA=0)
The variation of partial pressure of component B (pB) with its mole fraction (xB) is represented by the plot from xB =0 (i.e. pB= pB° × 0 = 0) to xB =1 (i.e. pB=pB°)
The total vapour pressure p, exerted by the solution as a whole at any composition is given by the sum of partial vapour pressures according to Dalton’s law of partial pressures.
Thus
p= pA +pB
p= pA° xA + pB° xB
The vapour pressure of solutions of different compositions, lies between the vapour pressure of the pure components (pA° and pB°) and they lie on the straight line joining pA° and pB°.
The minimum value of p (total vapour pressure) is pA° and the maximum value is pB° assuming that the component A is less volatile than component B i.e pA° < pB°
(b) Vapour Pressure of Solution of Solid in Liquid
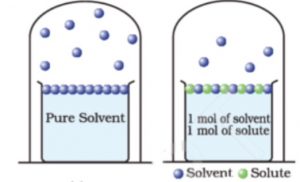
Add a small amount of a non-volatile solute to the solvent (e.g, sugar in water) to form the solution. When evaporation of this solution takes place, the vapour phase again consists of vapours of the solvent (i.e., of water) because the solute is non-volatile.The vapour pressure of the solution is found to be less than that of the pure solvent.
The vapour pressure depends on the escape of solvent molecules from the surface of the liquid.In the case of solution, the non-volatile sugar molecules occupy a certain surface area. As a result lesser number of solvent molecules will escape into vapours. Vapour pressure of the solution will be less than that of the pure solvent or there will be a lowering in vapour pressure. The increase in the concentration of sugar in the solution will further lower the vapour pressure of the solution.
Raoult’s law for liquid solutions containing non-volatile solutes
According to Raoult’s law the partial vapour pressure of volatile component in the solution is directly proportional to the mole fraction in it. When the solute is non-volatile, only the solvent molecules are present in the vapour phase. Therefore, the vapour pressure of the solution will be the vapour pressure due to solvent only.
If PA is the vapour pressure of the solvent over a solution containing non-volatile solute and xA is its mole fraction, then according to Raoult’s law, the vapour pressure of the solvent in the solution,
pA =pA° xA
p=pA°xA
p(solution) = p(pure solvent) × mole fraction of solvent
This relationship is known as Raoult’s law.
For solutions containing non-volatile solutes, the Raoult’s law may be stated as:
At a given temperature, the vapour pressure of a solution containing non-volatile solute is directly proportional to the mole fraction of the solvent.
If vapour pressure of the solution containing a non volatile solute is plotted against the mole fraction of the solvent, a straight line plot will be obtained as:
Rearranging the above equation, P/ PA° = xA
Subtracting each side of the equation from 1, we have
1- (p/pA°) = 1- xA
(pA°- p) / pA° = xB
(p°solvent -psolution) /p°solvent = xsolute
pA°- pA(difference in vapour pressure of pure solvent and solution)represents the lowering in vapour pressure on the formation of solution.
Thus, the Raoult’s law in its modified form may be stated as:
the relative lowering in vapour pressure of an ideal solution containing the non-volatile solute is equal to the mole fraction of the solute at a given temperature.
Raoult’s law as a special case of Henry’s law
The vapour pressure of a volatile component in given solution is given by the relation:
pA =PA° xA
where pA° is the vapour pressure of the pure component, pA is the vapour pressure in the solution having mole fraction xA. In the case of solution of a gas in a liquid, the gaseous component is volatile component. Its solubility is governed by Henry law which gives the relation:
p=KH x
where p is the pressure of the gas above the solution and x is its mole fraction ,KH is a proportionality constant known as Henry’s constant.
The partial pressure of the volatile component or gas is directly proportional to its mole fraction in solution. The only difference in the two expressions is the proportionality constant PA° (in Raoult’s law) and KH (in Henry’s law). Therefore, Raoult’s law becomes a special case of Henry’s law in which KH becomes equal to vapour pressure of the pure component (pA°).
Thanku so much helps a lot