1) Elements in which the last electron enters the s orbital are called s-block elements. Since s-subshell has only one orbital which can accommodate only two electrons, therefore there are only two groups of s-block elements.
2) Elements of group 1 (alkali metals) and group 2 (alkaline earth metals) constitute s- block elements. The elements of these groups contain one or two electrons in the outer most s-orbital while all the inner shells are completely filled.
3) Electronic configuration of group 1 may be represented as [Noble gas] ns1 while that of group 2(alkaline earth metals) may be represented as [Noble gas] ns2 Where n is the number of outermost shell.
4) Both alkali and alkaline earth metals are highly reactive and hence do not occur in the free state but occur in nature in the combined state. Alkali metals mostly occur as halides ,oxides ,silicates, borates and nitrates whereas alkaline earth metals mainly occur as silicates ,carbonates ,sulphates and phosphates.
5) Alkali metals have similar chemical properties yet they do not occur together mainly because their ions are of different sizes.
6) Some alkali and alkaline earth metals occur abundantly in nature. Calcium is the fifth, magnesium is the sixth, sodium is the seventh and potassium is the eighth, barium is the 14th and Strontium is the 15th most abundant element by weight in the earth crust. Sodium and magnesium are also present in relatively large amount in sea water, brine Wells and few salt lakes.
Anomalous Behaviour of First element of a group
The first element of a group differs considerable from the rest of the elements of the same group. This anomalous behaviour is due to a) small atomic and ionic radii b) high electronegativity and ionization enthalpy c) high polarising power of its cation d) absence of d- electron in its valence shell. Lithium differ from rest of the alkali metals and Be differs from rest of the alkaline earth metals.
Diagonal relationship
Some elements of certain groups in the second period resemble with the certain elements of the next higher group in the third period. This is called diagonal relationship.
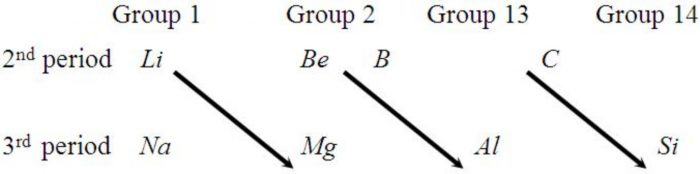
1) Similarity in electropositive character: Electronegativity increases along a period but decreases down a group.Electropositive character decreases along a period but increases down a group. On moving diagonally, these two opposing trends partly cancel out. As a result, diagonally related elements have similar electropositive character and hence exhibit similar properties.
2)Similarity in atomic or ionic radii : Atomic and Ionic radii decreases across a period but increases down a group. On moving diagonally these two trends partly cancel out. As a result, diagonally related elements have similar atomic and ionic radii and hence have similar properties.
3) Similarities in polarising power: On moving across a period ,the charge on the ion increases and size decreases ,causing the polarising power to increase. On moving down a group, the size increases and the polarising power decreases. On moving diagonally ,these to trends partly cancel out. As a result diagonally related elements have similar polarising power and hence have similar properties.
Sir please give me detail notes of chemistry class 11
This notes are useful for last mint. Revision thank you .