Question 1 What are colloids. Give example?
Question 2 What do you mean by Tyndall effect?
Question 3 Give properties of colloids?
Question 4 What are sol.Give example?
Question 5 What are aerosol.Give example?
Question 6 What are emulsions.Give example?
Question 7 What are foam.Give example?
Question 8 What is the difference between solution and colloids?
Colloids
A solution in which the size of solute particles is intermediate between those in true solution and suspension is called as Colloids.
For Example: Soap Solution,Starch solution,milk,Blood,ink etc.
Properties of Colloids
1) They appear to be homogeneous but actually they are heterogeneous when observed under microscope.
2) They are not perfectly transparent,it is somewhat translucent.
3) The particles cannot be seen by naked eyes or microscope.
4) The particles do not settle on keeping.
5) The particles cannot be separated by filtration.
6) The particles are big enough to scatter light.
7) The size of particles is bigger than those of true solution but smaller than suspension.
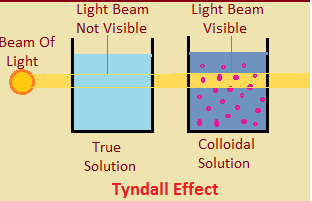
If a beam of light is put on a colloidal solution,kept in a beaker in a dark room,path of light beam is illuminated and become visible when seen from the side.
The path of light beam becomes visible because colloidal particles are big enough to scatter light falling on them in all direction.
The scattering of light by colloidal particles is called TYNDALL EFFECT.
A true solution does not scatter a beam of light passing through it but colloidal solution scatter a beam of light.
Types of Colloids
| Name of Colloid | Dispersed Phase | Dispersed Medium | Example |
| 1) Sol | Solid | Liquid | Ink, Soap Solution,Paints |
| 2) Solid Sol | Solid | Solid | Coloured Gemstones |
| 3) Aerosol | a)Solid
b)Liquid |
Gas
Gas |
Smoke,
Automobile exhaust Fog,Mist,Cloud |
| 4) Emulsions | Liquid | Liquid | Milk,Butter,
Cream |
| 5) Foam | Gas | Liquid | Shaving Cream,Soap Bubbles |
| 6) Solid Foam | Gas | Solid | Insulating Foam,Foam Rubber,sponge |
| 7) Gel | Liquid | Solid | Jellies,Gelatin |
This is very helpful for us ,now I understand my all doubts is clear.
THANKYOU
VERY HELPFUL!! THANKYOU SO SO MUCH!!
nice and cool explaination
Mam THANK YOU soo much.
You helped me in writing class work.
Once again THANK YOU mam.
That definition is very helpful for me so, thanks man thank you so much
Very nice and easy answers
yes it was really helpful, keep up the good work
Thanks for the notes and you all are doing great job by uploading notes very helpful
Thanks for helping me to complete my project this helped me a lot.