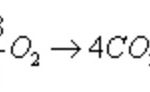Heat of reaction or enthalpy of reaction is a term used for the heat changes accompanying any reaction. Enthalpy of combustion The enthalpy of combustion of a substance is defined as the heat change when 1 mole of substance is completely burnt or oxidised in oxygen. CH4 ( g ) + 2O2 ( g ) ----------------> CO2 ( g ) + 2H2O ( g ) ΔcH= -890.4 KJ mol -1 Reaction … [Read more...] about Enthalpies of Reaction