Class 9 Science Chapter 1
Matter in Our Surroundings NCERT Solutions
Question Answers, Page 3
1. Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold drinks , lemon, water, smell of perfume.
Answer:
Anything which occupies space and has mass is called matter. There are three states of matter: solid, liquid and gas.
Chair, almonds, water, air, cold drink are matter.
2. Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer:
Solid diffuses at a very slow rate. If the temperature is increased then rate of diffusion of solid particles also increases. This is due to increase in kinetic energy of solid particles that the smell of hot sizzling food reaches us several meters away but to get the smell from cold food you have to go close.
3. A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer:
A diver is able to cut through water in a swimming pool show that matter is made up of particles and inter molecular spaces between the particles is weak.
4. What are the characteristics of the particles of matter?
Answer:
1)The particles of matter are very very small in size.
2)The particles of matter have spaces between them
3)The particles of matter are constantly moving
4)Particles of matter attract each other
Question Answers, Page 6
1. The mass per unit volume of a substance is called density. (density = mass/volume).
Arrange the following in order of increasing density – air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Answer: Air < Exhaust from chimney< Cotton< Water < honey < Chalk < Iron
2. (a) Tabulate the differences in the characteristics of states of matter.
Answer:
| Solid | Liquid | Gases |
| 1)They have fixed shape and volume | They do not have fixed shape but have fixed volume | They do not have fixed shape and volume |
| 2)They cannot be compressed | They cannot be compressed | They can be compressed easily |
| 3)They have high density | They have moderate density | They have low density |
| 4)They do not flow | They flow easily | They flow easily |
| 5)They do not fill their container | They do not fill their container | They fill their container |
| 6)The forces of attraction are strong | The forces of attraction are less strong than solids | The forces of attraction are weak. |
| 7)Kinetic energy is least | Kinetic energy is more than solids | Kinetic energy is maximum. |
| 8)Particles are closely packed | Particles are not close as in solids | Particles are much farther apart from one another. |
| 9)For Ex: Chair, table, chalk, book | For Ex: Water, petrol, cold drinks | For Ex: Oxygen, nitrogen, helium |
(b) Comment upon the following: rigidity, compressibility, fluidity, filling a gas
container, shape, kinetic energy and density.
Answer:
Rigidity is defined as the tendency of matter to resist a change in shape.
Compressibility is the ability to be reduced to a lower volume when force is
applied.
Fluidity is the ability to flow.
By filling a gas container we mean the attainment of shape of the container by
gas.
Shape defines a definite boundary.
Kinetic energy is the energy possessed by a particle due to its motion.
Density is mass per unit volume.
3. Give reasons
(a) A gas fills completely the vessel in which it is kept.
Answer:
Intermolecular forces of attraction between the gas molecules is negligible. Thus , gas particles move in all directions. Therefore gas completely fills the vessel in which
it is kept.
(b) A gas exerts pressure on the walls of the container.
Answer:
The intermolecular forces of attraction are weak in gases and therefore particles move randomly in all directions at high speed. As a result, the particles hit each other and also hit the walls of the container with a force. Therefore, gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
Answer:
A wooden table has a definite shape and volume. It is very rigid and cannot be compressed i.e., it has the characteristics of a solid. Hence, a wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood
we need a karate expert.
Answer:
Particles of air have large spaces between them. On the other hand, wood has little space between its particles. Also, it is rigid. For this reason, we can easily move our hands in air, but to do the same through a solid block of wood, we need a karate expert.
4. Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
Answer:
Density is defined as mass per unit volume. As the volume of substance increases , its density decreases. Though ice is a solid, it has large number of empty spaces between its particles. These spaces are larger as compared to the spaces present between the particles of water. Thus, the volume of ice is greater than that of water. Hence, the density of ice is less than that of water. A substance with lower density than water can float on water. Therefore, ice floats on water.
Question Answers, Page 9
1. Convert the following temperature to celsius scale:
a.) 300 K
b.) 573 K.
a) 300 K = (300 − 273)°C = 27°C
b) 573 K = (573 − 273)°C = 300°C
2. What is the physical state of water at:
a.) 250ºC
b.) 100ºC ?
a) Water at 250°C exists in gaseous state.
b) At 100°C, water can exist in both liquid and gaseous form.
3. For any substance, why does the temperature remain constant during the change of state?
Answer:
During the change of state, the substance converts itself from one form of physical state to another. When it is being converted, the energy supplied are utilised to form or break the intermolecular bonds thereby keeping the temperature of the substance constant.
4. Suggest a method to liquefy atmospheric gases.
Answer:
Atmospheric gases can be liquified by increasing pressure and decreasing temperature.
Question Answers, Page 10
1. Why does a desert cooler cool better on a hot dry day?
Answer:
A desert cooler cools better on a hot and dry day because the higher temperature on a hot day increases the rate of evaporation of water, and dryness of air also increases the rate of evaporation of water. And due to increased rate of evaporation of water, a desert cooler cools better on a hot and dry day.
2. How does the water kept in an earthen pot (matka) become cool during summer?
Answer:
During hot summer days, water is usually kept in an earthen pot to keep it cool. The earthen pot has a large number of extremely small pores in its walls. Some of the water continuously keeps seeping through these pores to the outside of the pot. This water evaporates continuously and takes the latent heat required for vaporisation from the earthen pot and remaining water. In this way, the remaining water loses heat and gets cooled.
3. Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Answer:
If we put a little of acetone or petrol or perfume on our palm and wave it around, the acetone or petrol or perfume evaporates rapidly and our hand feels very cool. This is due to the fact that to change liquid to vapour state, acetone or petrol or perfume requires latent heat of vaporisation. The acetone or petrol or perfume takes this heat of vaporisation from our hand. The hand loses heat and gets cooled.
4. Why are we able to sip hot tea or milk faster from a saucer rather than a cup?
Answer:
We are able to sip hot tea faster from a saucer than from a cup. This is because saucer has large surface area. Due to the large surface area, the evaporation of hot tea from saucer is faster.
5. What type of clothes should we wear in summer?
Answer:
We should wear cotton clothes in hot summer days to keep cool and comfortable. We get lot of sweat on our body in hot summer days. Now cotton is a good absorber of water, so it absorbs the sweat from our body and expose it to the air for evaporation. The evaporation of this sweat cools our body.
Exercise Page 12
1.Convert the following temperatures to the celsius scale.
(a) 293 K (b) 573 K.
Answer
a) 300 K = (300-273) = 27∘ C
b) 573 K= (573-273) = 300∘ C
2. Convert the following temperatures to the kelvin scale.
(a) 25°C (b) 373°C.
a) 25∘ C = 25+ 273 = 298 K
b) 373∘C = 373 + 273 = 646 K
3. Give reason for the following observations.
(a) Naphthalene balls disappear with time without leaving
any solid.
Answer : Naphthalene balls disappear with time without leaving any solid due to the process of sublimation.
(b) We can get the smell of perfume sitting several metres away.
Answer:
We can get the smell of perfume sitting several meters away due to diffusion. It is the property of matter which is based on the motion of particles. Vapours of perfume diffuse very fast.
4. Arrange the following substances in increasing order of forces of attraction between the particles— water, sugar, oxygen.
Oxygen< water < sugar
5. What is the physical state of water at—
(a) 25°C
Water is present in liquid state.
(b) 0°C
Water can exist both as solid and liquid.
(c) 100°C ?
Water can exist both as liquid and vapours
6. Give two reasons to justify—
(a) water at room temperature is a liquid.
Answer:
At room temperature water is a liquid because it does not have fixed shape , fixed volume and it can flow easily.
(b) an iron almirah is a solid at room temperature.
Answer:
An iron almirah is a solid at room temperature because it has fixed shape, fixed volume and high density.
7. Why is ice at 273 K more effective in cooling than water at the same temperature?
Answer:
At 273 K ice is more effective in cooling than water because at this temperature ice has less energy. Water possesses the additional latent heat of fusion.
8. What produces more severe burns, boiling water or steam?
Answer:
Steam has more latent heat of vaporisation and therefore it will produce more burns than boiling water.
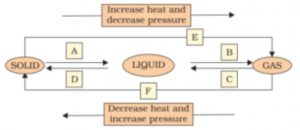
9. Name A,B,C,D,E and F in the following diagram showing change in its state
Answer:
A——–> Melting
B——–> Boiling or vaporisation
C———> Condensation
D———> Solidification
E———> Sublimation
F———> Sublimation
Leave a Reply